Building a thriving environment for medicine discovery
How are medicines and vaccines discovered?
The UK has a long-standing reputation for research into medicine and advances in basic science mean we understand diseases better than we've ever done before.
Dr Bryan Deane talks us through how the process of researching and developing a new medicine gets started.

What are clinical trials and how do they work?
Clinical trials are very important for finding out whether a medicine works and is safe. Every medicine used in the NHS has to be tested in a clinical trial before its used.
Dr Sheuli Porkess talks us through how potential new medicines and vaccines go through rigorous clinical trials.

In this #ValuingMedicine video Dr Emily Travis tell us how to know whether a clinical trial is right for you.
Patients involved in clinical research are vital to finding new treatments. Dr Emily Travis takes us through how, as a patient, you can take charge of your care in research #ValuingMedicine
How do we monitor and regulate medicines?
Clinical trials are essential for testing whether a medicine is safe and effective, but really its only the first step. They need to be monitored and regulated for their whole lifetime.
Clinical trials are essential for testing whether a medicine is safe and effective, but really its only the first step. They need to be monitored and regulated for their whole lifetime. Steve Hoare shows us who pharmaceutical companies continue to make sure a medicine is safe. and how they use real world data to advance research and discovery.
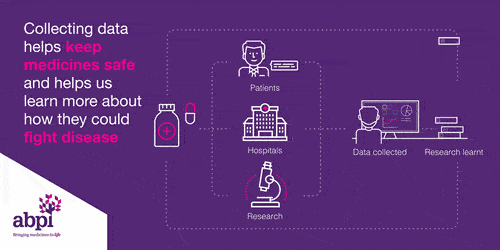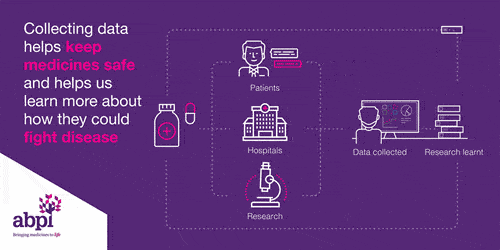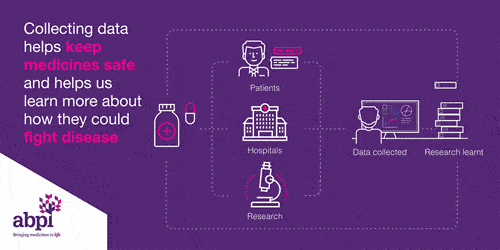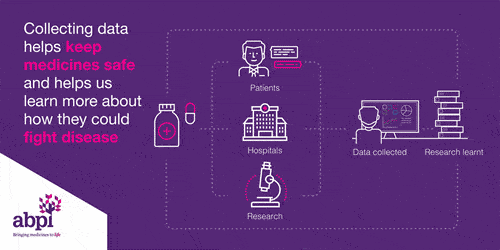
Clinical trials are very important for finding out if a medicine works and it is safe. Every medicine used in the NHS has to be tested before it can be used.
Clinical trials start with health volunteers and this will be the first time the medicine will be used in people. The next step is then to involve patients with the disease to see if the medicine works: does it delay the disease or cure the disease, and it is safe?
Before anyone gets involved with clinical trials they’ll sit down with the team and work out exactly what’s going to happen, any of the risks and whether a clinical trial is the right thing for them.
As we get more information the trials grow, with more patients in more hospitals not just in the UK but around the world. Trials can take anywhere from weeks to years depending on the type of disease and the type of medicine that we’re studying.
This information is share with the regulator who works out whether to grant a license or not.
New data and technology mean we can do research in new ways, looking into treatments that previously seemed impossible.
The NHS is very active in research with 700,000 people taking part in research for things like cancer, heart disease and diabetes.
Clinical trials are key for how we develop medicines of the future, but we can’t do it without patients and we’re hugely grateful for everyone that makes them happen.
How do we regulate medicines?
Clinical trials are really just the beginning of a medicines journey: they must be monitored and regulated for the rest of their lifetime.
A medicine’s license is the green light for it to be given to patients and it covers everything from what it should be used for, who it should be given to and what dose is appropriate.
The next step is to past strict tests to make sure its cost effective for the NHS.
Once a medicine is made available, the pharmaceutical company has a duty to monitor it and make sure it performs safely for as long as it’s on the market.
This process if called pharmacovigilance and it helps inform regulators about previously unknown side effects – it can also help collected real world data that can be fed back to improve future research.
More data means a better understanding of what a medicine can do – often meaning we can explore using it to treat other conditions or used in other sets of patients.
It may also be fed back to unlock basic science and help progress new treatments for future generations.
What technologies and new treatments are on the horizon?
The way we look for new treatments and cures is evolving, and medicines are more personalised now than ever before. Treatments are becoming more sophisticated as we gain a better understanding of science and disease.
The way we look for new treatments and cures is evolving, and medicines are more personalised now than ever before. Treatments are becoming more sophisticated as we gain a better understanding of science and disease. Dr Ali Hansford looks at what the future hold for medicines research and the treatments on the horizon.

Developments such as genomics are helping us to spot how diseases like cancer and affecting different patients differently. They’re giving us new clues for how to develop new medicines.
The first step is to identify the target; maybe a cell receptor or an enzyme or a gene that’s involved in the disease process. The next challenge is to find something which affects that target to interfere with or prevent the development of disease or symptoms.
We sometimes have to test tens of thousands of molecules either through computer modelling, or in test tubes or cell cultures or in animals to identify a few that are effective enough to take forward to trial in patients.
The biopharmaceutical industry invests more than anyone else in research and development.
New science is becoming more and more precise. Despite the fact that failure rates are still high we’re learning about disease all the time and when we do have successes, we’re bringing improvements to the lives of millions of patients around the world.
Who do we need to make the medicines of the future?
Pharmaceutical companies around the world are pioneering new medicines and vaccines. The UK's life sciences sector employs over 250,000 people alone
Pharmaceutical companies around the world are pioneering new medicines and vaccines. The UK's life sciences sector employs over 250,000 people alone. Andrew Croydon looks at what skills we need to develop the medicines of the future, and the skills gaps we need to fill if we're going to keep the UK ahead.

Last modified: 09 May 2024
Last reviewed: 09 May 2024